
Commonwealth of Virginia
Office of the Attorney General
|
Mark Herring |
202 North Ninth Street |
For media inquiries only, contact:
Charlotte Gomer, Press Secretary
Phone: (804)786-1022
Mobile: (804) 512-2552
Email:
UNSEALED GENERIC DRUG COMPLAINT SHOWS EXTENT OF CONSPIRACY TO FIX PRICES
RICHMOND (June 26, 2019) – Attorney General Mark R. Herring joined Connecticut Attorney General William Tong and 41 other states today in releasing the full, unredacted complaint against Teva Pharmaceuticals and 19 of the nation's largest generic drug manufacturers after the court granted the states' motion to unseal the complaint. Among the evidence now public are emails between generic drug manufacturers coordinating their response to a Congressional inquiry, emails enforcing "fair share” and "playing nice in the sandbox” market allocation, "fluff pricing” strategy and other brazen coordination to artificially inflate prices, hinder competition and unreasonably restrain trade across the industry. The lawsuit was first filed last month in U.S. District Court for the District of Connecticut.
"The allegations in this unsealed complaint show the shocking lengths that these drug manufacturers went to in order to hide their price fixing plans, mislead Congress and even destroy evidence,” said Attorney General Herring. "What these companies did – inflating generic drug prices and forcing hardworking people to pay much more for the drugs that they rely on – is unconscionable. The public needs to see just how brazenly these companies worked to line their own pockets without any regard for the people they would be harming in the process.”
"Polite F-U Letters”
In early October 2014, Heritage Pharmaceuticals received a letter from Congressman Elijah Cummings and Senator Bernie Sanders as part of their joint investigation into price increases in the generic drug industry. Now emails unsealed by the court show that Heritage outside counsel immediately coordinated a response with counsel for Teva and Mylan.

"No emails please”
Executives knew their behavior was illegal, and they sought to cover their tracks by limiting communication in writing. Still, ample written evidence of their conspiracy has now been released that show consciousness of guilt.

(pg. 50)
"I guess this is what they call co-opetition”
In one series of exchanges, senior executives at Mylan and Sandoz allegedly colluded to divvy up market share for a blood pressure medication. Both companies were the first to launch generic Valsartan on the same day, September 12, 2012. Leading up to the launch, records show company representatives spoke at least 21 times by phone to divvy up the market so that each competitor could obtain roughly 50 percent market share. The conspiracy apparently pleased company executives, including a Sandoz executive who states, "sometimes a little help from our competition is welcome as well.” Another senior executive replied: "I guess this is what they call 'co-opetition.”

High Quality Competitors
As Director of National Accounts at Teva, Defendant Nisha Patel's primary responsibility was to implement price increases. The complaint alleges that she did this by systematically conspiring with Teva's competitors and maintained a ranking system of Teva's competitors based on their collusive relationships, with +3 assigned to the most collusive and -3 assigned to the least. Detailed rankings and charts documenting her communication with competitors have been unsealed.

(pg. 176)

(pg. 183)
Industry Code Words Revealed: Playing Nice in the Sandbox, Fair Share, Fluff Pricing
The unredacted complaint reveals commonly used code words used by coconspirators as they colluded with competitors to divvy up market share and coordinate on price increases in violation of federal antitrust law. These include "fair share”, "playing nice in the sandbox”, and "fluff pricing”.


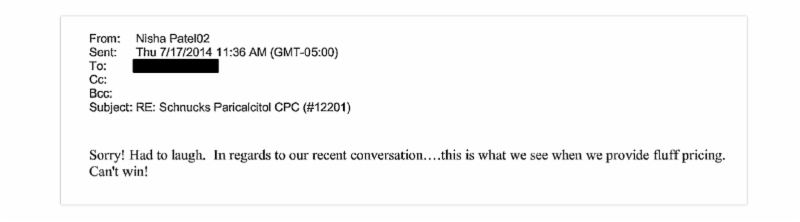
(pg. 147)
The complaint is the second to be filed in an ongoing, expanding investigation that the Connecticut Office of the Attorney General has referred to as possibly the largest cartel case in the history of the United States. The first complaint, still pending in U.S. District Court in the Eastern District of Pennsylvania, was filed in 2016 and now includes 18 corporate defendants, two individual defendants, and 15 generic drugs. Two former executives from Heritage Pharmaceuticals, Jeffery Glazer and Jason Malek, have entered into settlement agreements and are cooperating with the Attorneys General working group in that case.
In addition to Virginia, Alabama, Alaska, Arizona, Colorado, Connecticut, Delaware, Florida, Georgia, Hawaii, Idaho, Illinois, Indiana, Iowa, Kansas, Kentucky, Louisiana, Maine, Maryland, Massachusetts, Michigan, Minnesota, Mississippi, Missouri, Montana, Nebraska, Nevada, New Jersey, New Mexico, New York, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, Rhode Island, South Carolina, Tennessee, Utah, Vermont, Washington, West Virginia, Wisconsin and Puerto Rico joined the suit.
# # #